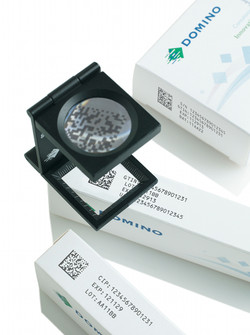
Viewpoint: A shot in the arm for generics producers
Domino inksThere is nowhere to hide from the Falsified Medicines Directive. It impacts generics producers in the same way as any other drug manufacturer or re-packager selling into Europe. With just under 48 months to go before the deadline, which is likely to be mid-2018, the clock is ticking for all producers to get their house in order.
The subject was hot on the agenda at the European Generics Association (EGA) meeting in Madrid earlier this month. As generics manufacturers often supply products with much lower margins than patented drug producers, their budgets are restricted when it comes to new investment – which is why many of them have been holding out until the last minute to find the ‘best-fit’ solution for their businesses.
Related Posts
NASCAR News: Derek White suspended for alleged smuggling involvement
Xfinity Series owner and driver Derek White has been suspended indefinitely by...
UP ATS nabs 4th accused in inter-state fake currency racket; Rs 1.05 lakh seized from Sitapur man; gang used Alibaba-sourced paper, Photoshop files to mimic notes
The UP Anti-Terrorist Squad (ATS) has apprehended a fourth suspect, Anas Ahmad,...
Fake Tata wire brand products seized in Andhra Pradesh
The Tata Steel intelligence unit along with Andhra Pradesh police seized...
Declared as oranges to evade tax, 1.25cr cigarettes seized in Ctg
Chattogram Custom House officials seized 1.25 crore cigarette sticks that were...






